While many cis-NATs have been well-characterized among prokaryotes, relatively little is known about the role of cis-NATs in eukaryotes. Recent reports have suggested that cis-NATs occur relatively frequently in human and other mammalian genomes, which came as a surprise considering the low gene density of these genomes compared to prokaryotes. To date, more than 2000 human cis-NATs, 2400 mouse cis-NATs and >1000 drosophila cis-NATs were predicted in the respective genomes. Comparative analyses comparing the conservation of cis-NATs among various species (e.g. human, mouse, fugu) are still in the infancy stages and have yet to arrive at any meaningful conclusions. Notably, tail-to-tail overlapping pairs of sense-antisense RNAs were more common than head-to-head pairings, emphasizing the importance of the RNA 3’ region in post-transcriptional regulation. In addition, only 6.6% of human sense-antisense RNA pairs are found in mouse, of which most (95.6%) involves two protein coding genes, suggesting that cis-NAT-mediated regulation may only be crucial for species-specific cellular processes. (Numata, 2007)
Due to the diverse roles attributed to cis-NATs, there is no significant partner RNA or protein that consistently plays a role in cis-NAT function. For instance, protein machinery like Dicer involved in the RNAi pathway will be utilized when a particular cis-NAT downregulates its sense strand partner by RNAi.
Cis-NATs exert their effects on cellular function via a wide variety of mechanisms. A few examples are listed below to highlight the many distinct modes of action involved.
Prokaryotic
- Transcription attentuation
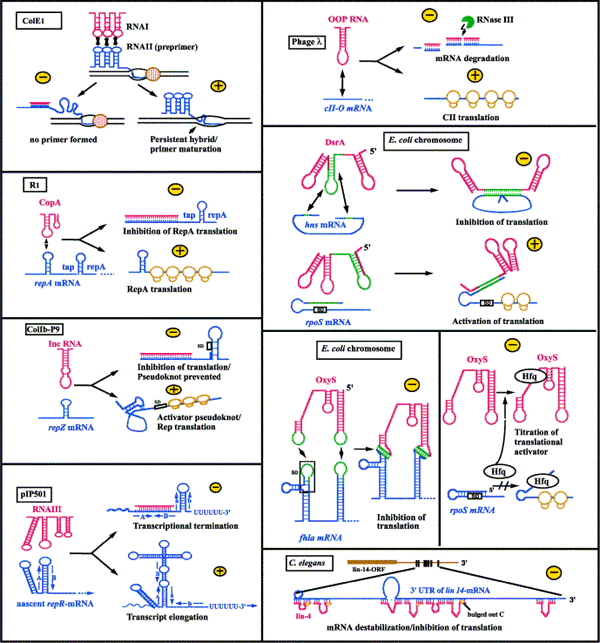
The rep gene in staphylococcal plasmids pT181, pIP501 and pAMß1 encodes a protein essential for origin recognition and plasmid replication. In the presence of the antisense rep RNA, the sense rep RNA changes secondary structure to include a terminator stem-loop structure upstream of the Shine-Dalgarno sequence, thereby preventing rep protein synthesis. The antisense RNA exerts its inhibitory effects without the help of any helper protein. - Translation inhibition
Several cis-NATs have been shown to inhibit translation by direct binding to the ribosomal binding site of the sense RNA. Some examples of this inhibition mechanism include: RNAII in plasmid pLS1 which blocks repB translation, FinP of plasmids F and R1 which blocks traJ translation. - Regulation of plasmid copy number via inhibition of primer maturation
ColE1 is a small, multicopy plasmid whose copy number is tightly self-regulated in the bacteria. Plasmid replication is initiated by the transcription of a RNA primer, RNA II, which lies 555 nt upstream of the origin of replication (ori) site. When RNA polymerase nears the origin during transcription, the RNA transcript forms a persistent hybrid with the template DNA. This hybrid is cleaved by RNase H, and the resulting 3' end serves as a primer for DNA synthesis by DNA polymerase and subsequent plasmid replication. RNA I is an antisense RNA which shares sequence complementarity with RNA II, and is transcribed from the same plasmid region as RNA II but via a different promoter. RNA I is constitutively synthesized and rapidly turned over, such that its intracellular concentration correlates with plasmid copy number at any time during the cell cycle. As such, when high levels of the ColE1 plasmid are present in the cell, RNA I is highly expressed, RNA I binds to RNA II, prevents the formation of the persistent hybrid and inhibits plasmid replication. - Prevention of formation of an activator RNA pseudoknot
Rep expression in IncB, IncIa, IncK, IncL and IncM plasmids involves a long-distance activator RNA pseudoknot. A leader peptide ORF, repY, must be translated in order to enable RepZ synthesis to disrupt an inhibitory stem-loop at the rep ribosomal binding site. This permits formation of a short helix between the target loop and disrupted stem, located 100 nt apart. This long-distance pseudoknot activates repZ translation. The corresponding antisense RNAs have a dual function: they block leader peptide translation and pseudoknot formation. - Promotion or inhibition of mRNA degradation
While most of the cis-NATs expressed by plasmids have unique mechanisms of action, nearly all known cis-NATs encoded by the bacterial chromosome appear to affect either translation or mRNA stability. The detailed mechanism involved with this form of regulation has not been elucidated. Strongest evidence comes from experimental data for the RatA/TxpA antitoxin/toxin system from Bacillus subtilis in support of antisense RNA mediated mRNA degradation, although an involvement of RNase III is still elusive. Other examples include the post-transcriptional regulation of ldrD toxin gene by RdlD antitoxin RNA in E. coli.
Eukaryotic
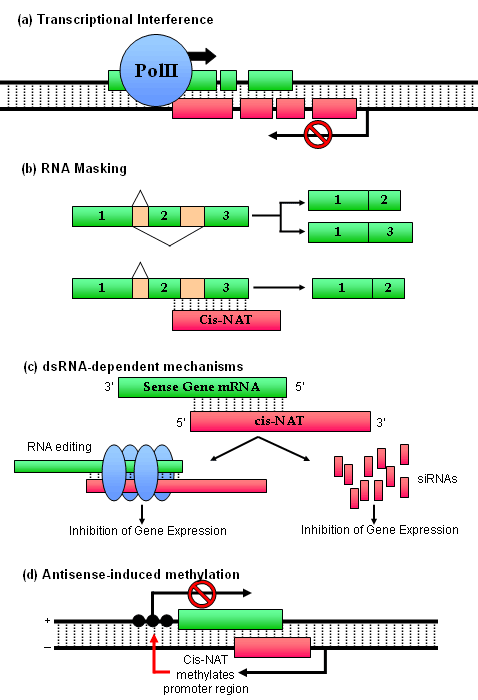
- Transcriptional interference
As transcription involves large protein complexes and the unwinding of local duplex DNA, it is unlikely for two overlapping transcriptional units to be transcribed simultaneously. As such, one hypothesis on the mechanism of action of cis-NATs is the downregulation of target sense gene transcription by competitive transcriptional interference from the corresponding cis-NAT. This is supported by the finding that some sense-antisense pairs exhibit reciprocal expression. For example, transcription of initiation factor 2a (eIF2a) and its associated cis-NAT are inversely correlated at different stages of the cell cycle, and deletion/mutation of the antisense RNA promoter increases sense RNA expression (Silverman, 1992, Prescott, 2002). - RNA masking
RNA duplex formation from the complementary binding between sense and antisense transcripts may mask critical regulatory features within the transcripts, thereby inhibiting the binding of other trans-acting factors. This mechanism could potentially regulate any step in gene expression, including mRNA splicing, transport, polyadenylation, translation and degradation. One example is the inhibition of alternative splicing by the cis-NAT Rev-ErbAa, which shares sequence complementarity with one of two functionally antagonistic splice forms of the thyroid hormone receptor ErbAa mRNA (ErbAa2 not ErbAa1) (Munroe, 1991). In different B-cell lines, expression of Rev-ErbAa correlates with an increase in the ratio of ErbAa1- to ErbAa2-mRNA levels. In addition, expression of Rev-ErbAa inhibits production of ErbAa2 from an ErbAa minigene in cells, and antisense RNAs complementary to the ErbAa2-specific exon efficiently and specifically block ErbAa2 splicing in vitro. These results show that a cis-NAT can specifically inhibit the alternative splicing of an mRNA, probably by blocking the accessibility of cis regulatory elements in the RNA. - dsRNA-dependent mechanisms and RNA interference
In addition to RNA masking, formation of double stranded RNAs may affect gene expression via RNA editing or RNAi gene slicing pathways. In the former, long perfect duplex of >100 bp are hyper-edited so that ~50% of adenosines on each strand are deaminated and results in nuclear retention or degradation of the RNA (Bass, 2002). In the latter case, the enzyme Dicer is known to cleave dsRNA into 21-23 nt duplexes and downregulate the corresponding RNA transcripts. - Antisense-induced methylation
Antisense transcription is known to affect gene expression but the underlying mechanism of action remains unclear. This is particularly true for antisense transcripts involved in gene imprinting or the methylation of CpG islands and chromatin remodeling. (See below for "antisense hemoglobin α-2 in α-thalassemia" for an example of this phenomenon.)
Cis-NATs have been found to regulate multiple processes in prokaryotes, such as replication initiation, conjugation efficiency, suicide, transposition, mRNA degradation and translation initiation. In eukaryotes, cis-NATs have been shown to regulate genomic imprinting, RNA interference, translational regulation, alternative splicing, X-inactivation and RNA editing. Thus far, cis-NATs have mainly been found to serve as negative regulators, either as gene dosage-dependent regulators or as secondary repressors of gene expression. While mechanisms of positive regulation are possible, only a few examples have been observed.
- Brantl S. Regulatory mechanisms employed by cis-encoded antisense RNAs. Curr Opin Microbiol. 2007 Apr;10(2):102-9. Pubmed, Article.
- Lavorgna G, Dahary D, Lehner B, Sorek R, Sanderson CM, Casari G. In search of antisense. Trends Biochem Sci. 2004 Feb;29(2):88-94. Pubmed, Article.
- Wagner EG, Simons RW. Antisense RNA control in bacteria, phages, and plasmids. Annu Rev Microbiol. 1994;48:713-42. Pubmed, Article.
- Werner A, Berdal A. Natural antisense transcripts: sound or silence? Physiol Genomics. 2005 Oct 17;23(2):125-31. Pubmed, Article.
- Bass BL. RNA editing by adenosine deaminases that act on RNA. Annu Rev Biochem. 2002;71:817-46. Pubmed, Article.
- Mihalich A, Reina M, Mangioni S, Ponti E, Alberti L, Viganò P, Vignali M, Di Blasio AM. Different basic fibroblast growth factor and fibroblast growth factor-antisense expression in eutopic endometrial stromal cells derived from women with and without endometriosis. J Clin Endocrinol Metab. 2003 Jun;88(6):2853-9. Pubmed, Article.
- Munroe SH, Lazar MA. Inhibition of c-erbA mRNA splicing by a naturally occurring antisense RNA. J Biol Chem. 1991 Nov 25;266(33):22083-6. Pubmed, Article.
- Numata K, Okada Y, Saito R, Kiyosawa H, Kanai A, Tomita M. Comparative analysis of cis-encoded antisense RNAs in eukaryotes. Gene. 2007 May 1;392(1-2):134-41. Pubmed, Article.
- Prescott EM, Proudfoot NJ. Transcriptional collision between convergent genes in budding yeast. Proc Natl Acad Sci U S A. 2002 Jun 25;99(13):8796-801. Pubmed, Article.
- Runte M, Hüttenhofer A, Gross S, Kiefmann M, Horsthemke B, Buiting K. The IC-SNURF-SNRPN transcript serves as a host for multiple small nucleolar RNA species and as an antisense RNA for UBE3A. Hum Mol Genet. 2001 Nov 1;10(23):2687-700. Pubmed, Article.
- Silverman TA, Noguchi M, Safer B. Role of sequences within the first intron in the regulation of expression of eukaryotic initiation factor 2 alpha. J Biol Chem. 1992 May 15;267(14):9738-42. Pubmed, Article.
- Spiegelman WG, Reichardt LF, Yaniv M, Heinemann SF, Kaiser AD, Eisen H. 1972. Bidirectional transcription and the regulation of phage ? repressor synthesis. Proc. Natl. Acad. Sci. USA 69:3156-60. Pubmed, Article.
- Tufarelli C, Stanley JA, Garrick D, Sharpe JA, Ayyub H, Wood WG, Higgs DR. Transcription of antisense RNA leading to gene silencing and methylation as a novel cause of human genetic disease. Nat Genet. 2003 Jun;34(2):157-65. Pubmed, Article.
- Zhang Y, Li J, Kong L, Gao G, Liu QR, Wei L. NATsDB: Natural Antisense Transcripts DataBase. Nucleic Acids Res. 2007 Jan;35(Database issue):D156-61. Pubmed, Article.
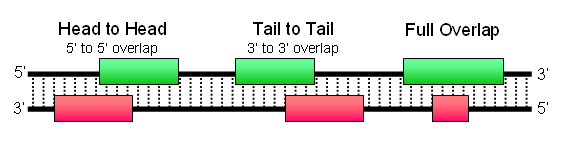
Cis-NATs have been assumed to be a typical PolII-dependent RNA transcript which utilizes the same transcriptional machinery as its sense strand partner for expression. By definition, cis-NATs are transcribed off the antisense strand of another gene and share at least partial sequence complementarity with their sense strand partner. There are three orientations in which this could happen, the most common of which is the tail-to-tail orientation where the 3’ ends of both transcripts align together, while head-to-head and complete overlap being less frequent.
Cis-NATs have transcriptional promoters independent of the sense gene to drive their own expression. Several cis-NATs have been shown to undergo splicing, nuclear export as well as protein expression. As the primary mechanism of action of cis-NATs varies greatly between the few known examples, the specific details of the biogenesis process are currently unknown.
Currently, cis-NATs are not widely used as molecular tools in biological research due to our lack of understanding of their mechanisms of action in various cellular contexts. However, as our understanding of their roles in regulating cellular function like sense strand expression improves, we can potentially mimic endogenous cis-NAT function via exogenous expression of specially-designed small RNAs which share sequence complementarity with our gene of interest.
Natural antisense transcripts, or NATs, are naturally occurring RNA transcripts which share sequence complementarity with other endogenous RNA transcripts. NATs can be broadly classified into two groups based on their locus of origin. Cis-natural antisense transcripts (cis-NATs) are transcripts which are transcribed at the same genomic loci of other genes, but from the opposite direction and off the opposite DNA strand. Cis-NATs have also been referred to as anti-parallel overlapping genes if both sense and antisense sequences are protein-coding genes. Trans-NATs, on the other hand, are transcribed from different genomic loci from the genes with which they share sequence complementarity and include small RNAs like siRNA and microRNA.
Non-overlapping antisense transcripts (NOT) form another closely-related but distinct class of antisense RNA transcripts which, similar to cis-NATs, are transcribed at the same genomic loci as another gene. However, NOTs are unique from cis-NATs because NOTs do not share sequence complementarity with the sense RNA due to the effects of splicing.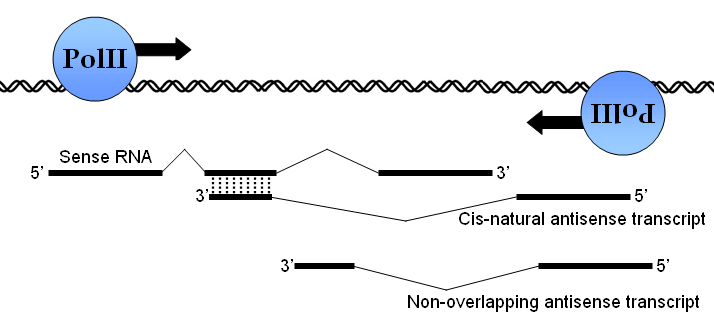
Regulation by cis-antisense RNA transcripts was first postulated in 1972 based on work in bacteriophage λ gene regulation. cI and cro are two essential genes coding for transcription inhibitors whose coding sequence lie adjacent to each other but in opposite directions. An alternative promoter for cI was discovered to be on the other side of cro, and initial studies confirmed the presence of cis-antisense cro RNA transcripts. This led the authors to hypothesize that this novel antisense RNA transcript might potentially serve a role in regulating cro gene activity (Spiegelman, 1972).
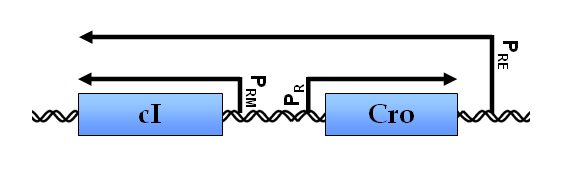
The functional significance of cis-NATs was not validated until a decade later when an antisense RNA, RNA I, was found to regulate the copy number of plasmid ColE1 by the inhibiting the maturation of a primer essential for DNA replication.
While most of the research conducted on cis-NATs has primarily focused on prokaryotes, the advent of genomic tools has uncovered new avenues to discovering cis-NATs in the eukaryotic genomes, including human, mouse, Drosophila, rice and Arabidopsis thaliana. Several approaches were used to identify these antisense transcripts, by either annotating cDNA clones to genome sequences of organisms of interest or by applying the BLAST algorithm between complete mRNA or EST sequences. In all, more than 2000 human cis-NATs, 2400 mouse cis-NATs and >1000 Drosophila cis-NATs were predicted to exist in the respective genomes.
Given the diverse regulatory functions and the widespread abundance of cis-NATs in the human genome, it came as no surprise when some cis-NATs were implicated in human diseases.
- Antisense UBE3A in Prader-Willi and Angelman syndromes
Human chromosome 15 contains a region of imprinted genes where gene expression is limited to one of the two homologous chromosomes and is causally linked to the Prader-Willi syndrome/Angelman syndrome (PWS/AS). PSW/AS is a genetic disease characterized by obesity, decreased muscle tone and decreased mental capacity, and results from a complete loss of these imprinted genes. One study reported the expression of a novel ~20kb antisense RNA that shares sequence complementarity with the UBE3A gene in the SNURF-SNRPN gene cluster, and as such hypothesized that paternally-expressed UBE3A antisense transcript could potentially regulate maternal expression of UBE3A and contribute to PWS/AS (Runte, 2001). - Antisense bFGF in endometriosis
Reduced expression of the exon 1B isoform of the basic fibroblast growth factor (bFGF) antisense transcript (1B FGF-AS) is correlated with the proliferation of endometrial cells observed in patients with endometriosis (Mihalich, 2003). - Antisense hemoglobin α-2 in a-thalassemia
α-thalassemia is an inherited autosomal recessive anemia that results from the underproduction of globin genes. One study described an individual with inherited α-thalassemia who has a chromosomal deletion that juxtaposes a structurally normal α-globin gene (HBA2) with a now-truncated, widely expressed downstream gene (LUC7L) that is transcribed in the opposite direction (Tufarelli, 2003). This results in the aberrant expression of an antisense HBA2 RNA transcript, which was shown to methylate the HBA2 CpG island and results in the transcriptional silencing of the intact gene.
